Recycling experiments with CAT-4 at 5 bar CO pressure (0.4 mmol 1a, 1.0... | Download Scientific Diagram
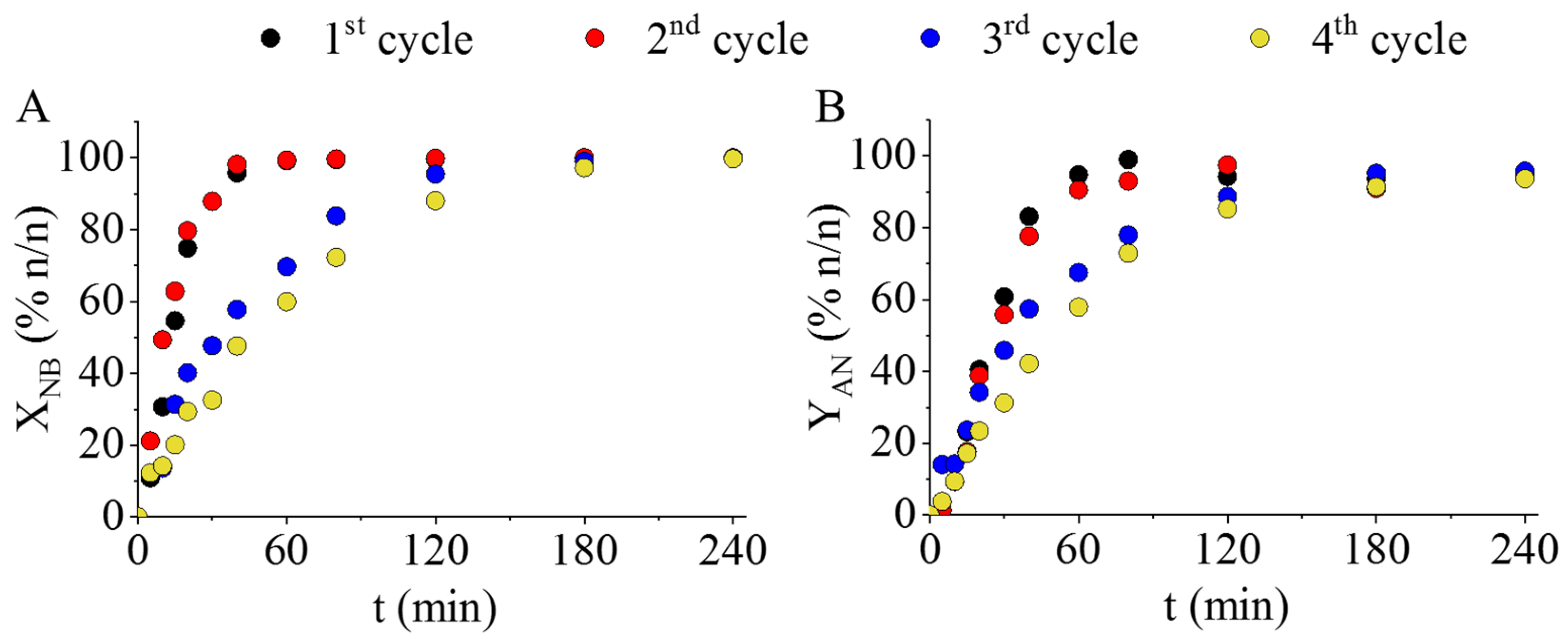
IJMS | Free Full-Text | Development of High-Efficiency, Magnetically Separable Palladium-Decorated Manganese-Ferrite Catalyst for Nitrobenzene Hydrogenation
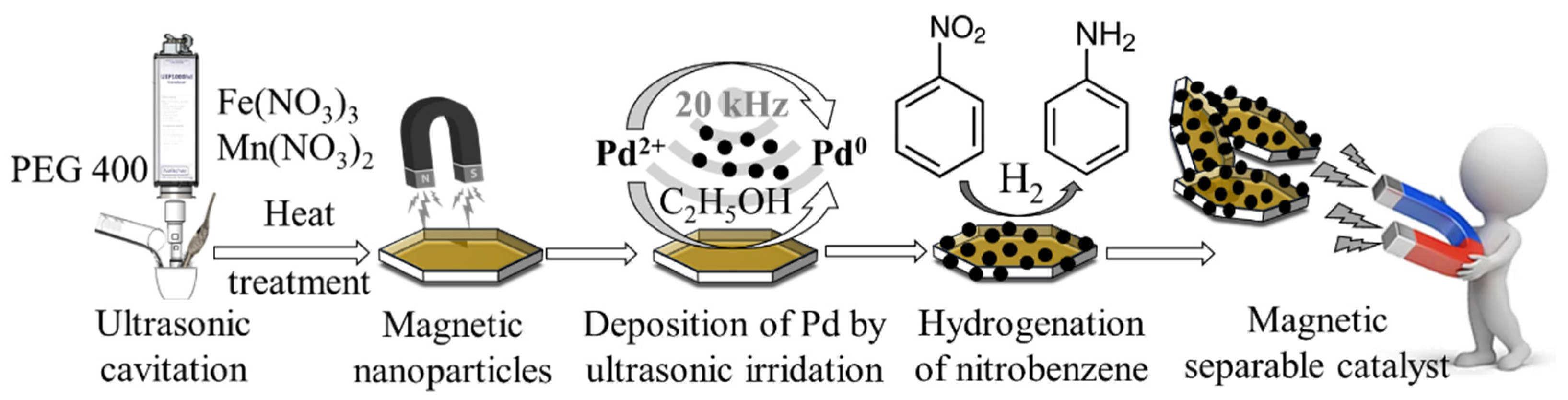
IJMS | Free Full-Text | Development of High-Efficiency, Magnetically Separable Palladium-Decorated Manganese-Ferrite Catalyst for Nitrobenzene Hydrogenation

Palladium bis(2,2,6,6‐tetramethyl‐3,5‐heptanedionate) catalyzed alkoxycarbonylation and aminocarbonylation reactions - Tambade - 2009 - Applied Organometallic Chemistry - Wiley Online Library

Synthesis of Dibenzopyranones through Palladium‐Catalyzed Directed C–H Activation/Carbonylation of 2‐Arylphenols - Luo - 2013 - Angewandte Chemie - Wiley Online Library

Molecules | Free Full-Text | Synthesis of Novel Cavitand Host Molecules via Palladium-Catalyzed Aryloxy- and Azidocarbonylation

Palladium‐Catalyzed Carbonylative Homocoupling of Aryl Iodides for the Synthesis of Symmetrical Diaryl Ketones with Formic Acid - Wu - 2018 - ChemCatChem - Wiley Online Library

PDF) Palladium nanoparticles on a pyridinium supported ionic liquid phase: a recyclable and low-leaching palladium catalyst for aminocarbonylation reactions †

Palladium nanoparticles embedded in MOF matrices: Catalytic activity and structural stability in iodobenzene methoxycarbonylation - ScienceDirect

Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics

Heterogenized Phenanthroline–Pd(2+)-Catalyzed Alkoxycarbonylation of Aryl Iodides in Base-Free Conditions | The Journal of Organic Chemistry
![Highly Chemoselective One‐Step Synthesis of Novel N‐Substituted‐Pyrrolo[3,4‐b]quinoline‐1,3‐diones via Palladium‐Catalyzed Aminocarbonylation/Carbonylative Cyclisation Sequence - Chniti - European Journal of Organic Chemistry - Wiley Online Library Highly Chemoselective One‐Step Synthesis of Novel N‐Substituted‐Pyrrolo[3,4‐b]quinoline‐1,3‐diones via Palladium‐Catalyzed Aminocarbonylation/Carbonylative Cyclisation Sequence - Chniti - European Journal of Organic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/f2bc5a20-ef7a-4b7a-a8b4-b4adedb597ec/ejoc202201374-toc-0001-m.jpg)
Highly Chemoselective One‐Step Synthesis of Novel N‐Substituted‐Pyrrolo[3,4‐b]quinoline‐1,3‐diones via Palladium‐Catalyzed Aminocarbonylation/Carbonylative Cyclisation Sequence - Chniti - European Journal of Organic Chemistry - Wiley Online Library












